Neurosurg Focus. 2012 Mar;32(3):E12.
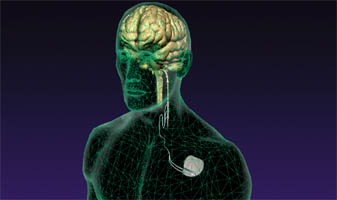
Vagal nerve stimulation for the treatment of medically refractory epilepsy: a review of the current literature.
Source
Department of Neurosurgery, Louisiana State University Health Sciences Center-Shreveport, Louisiana 71130-3932, USA.Abstract
OBJECT:
The authors conducted a study to evaluate the published results of vagal nerve stimulation (VNS) for medically refractory seizures according to evidence-based criteria.METHODS:
The authors performed a review of available literature published between 1980 and 2010. Inclusion criteria for articles included more than 10 patients evaluated, average follow-up of 1 or more years, inclusion of medically refractory epilepsy, and consistent preoperative surgical evaluation. Articles were divided into 4 classes of evidence according to criteria established by the American Academy of Neurology.RESULTS:
A total of 70 publications were reviewed, of which 20 were selected for review based on inclusion and exclusion criteria. There were 2 articles that provided Class I evidence, 7 that met criteria for Class II evidence, and 11 that provided Class III evidence. The majority of evidence supports VNS usage in partial epilepsy with a seizure reduction of 50% or more in the majority of cases and freedom from seizure in 6%-27% of patients who responded to stimulation. High stimulation with a gradual increase in VNS stimulation over the first 6 weeks to 3 months postoperatively is well supported by Class I and II data. Predictors of positive response included absence of bilateral interictal epileptiform activity and cortical malformations.CONCLUSIONS:
Vagal nerve stimulation is a safe and effective alternative for adult and pediatric populations with epilepsy refractory to medical and other surgical management.- PMID:
- 22380853
- [PubMed - indexed for MEDLINE]
http://www.ncbi.nlm.nih.gov/pubmed/22380853
Related citations in PubMed
- Refractory epilepsy in tuberous sclerosis: vagus nerve stimulation with or without subsequent resective surgery.[Epilepsy Behav. 2009]
- Review Vagus nerve stimulation for epilepsy: a meta-analysis of efficacy and predictors of response.[J Neurosurg. 2011]
- Final Report on Carcinogens Background Document for Styrene.[Rep Carcinog Backgr Doc. 2008]
- Vagus nerve stimulation for medically refractory epilepsy; efficacy and cost-benefit analysis.[Acta Neurochir (Wien). 1999]
- Review ILAE treatment guidelines: evidence-based analysis of antiepileptic drug efficacy and effectiveness as initial monotherapy for epileptic seizures and syndromes.[Epilepsia. 2006]