To the readers,
The reason I posted this article is the fact I use to suffer for years from debilitating cluster headaches.
Clinical Pain Medicine
ISSUE: OCTOBER 2015 | VOLUME: 13(10)
Noninvasive VNS Potentially Helpful for Acute Treatment of Cluster Headache
The results of the placebo-controlled ACT1 (Non-invasive Vagus Nerve Stimulation for the Acute Treatment) study, sponsored by electroCore, confirm that noninvasive vagus nerve stimulation (nVNS) may be a helpful acute intervention in selected patients with cluster headache (CH).
ElectroCore’s nVNS gammaCore device did not significantly increase the proportion of patients who had a pain-intensity scale score of 0 or 1 within 15 minutes of the start of therapy during their first CH attack. It also did not significantly lower average pain intensity at 15 minutes after treatment initiation, according to a presentation of the results at the American Headache Society’s 2015 Annual Scientific Meeting (abstract LBP07). Moreover, an accompanying poster indicated that true blinding was not achieved (abstract LBP08).
However, nVNS was associated with a higher proportion of sustained responders, defined as response at both 0.25 and one hour after treatment initiation. The respective percentages of sustained responders to nVNS or sham treatment were 26.7% and 12.3%.
“This is a doubling of the percentage of sustained responders, which we feel is clinically meaningful,” co-investigator Eric Liebler, BA, vice president, Scientific and Medical Affairs, electroCore, told Pain Medicine News in a telephone interview. “The patient wants to know whether their pain will go away or be much reduced, and they want it not only to go away, but to stay away.”
He said the company has conducted a similar study in Europe, and plans to submit an application that is based on data from both continents to the FDA for approval to market the device in the United States.
Nathaniel Schuster, MD, headache and facial pain fellow in the Department of Neurology at Montefiore Headache Center/Albert Einstein College of Medicine, in New York City, said he is looking forward to more studies of nVNS.
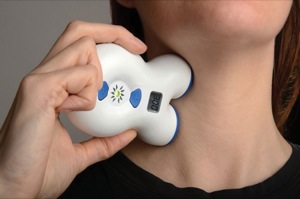
Photo: electroCore’s gammaCore device.
“I think that, if it proves to be effective, it will be welcomed eagerly by patients and physicians alike,” Dr. Schuster told Pain Medicine News. “Designing a sham control that effectively blinds trial subjects appears to be a challenge with this device, and a sham control with more effective blinding would improve the quality of the evidence.”
The ACT1 study initially included only people with episodic CH, but the investigators expanded it to also include those with chronic CH.
The sham device had an identical weight, visual and audio feedback, and user interface to the active nVNS device. However, the sham device generated a low-amplitude, low-frequency signal that did not stimulate the vagus nerve but produced a sensation by stimulating skin-touch receptors, according to Mr. Liebler. The active device produced a higher-amplitude, higher-frequency signal that stimulated the vagus nerve.
Seventy-three patients were randomly assigned to nVNS and another 77 to sham treatment for one month, followed by an open-label, three-month phase in which all patients received active nVNS. The two groups had similar demographic and CH characteristics.
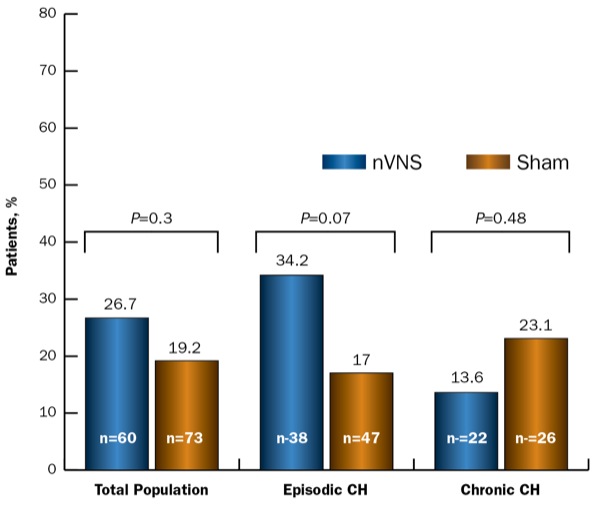
There was a 26.7% response rate at 15 minutes with active nVNS and a 19.2% 15-minute response rate with sham (P=NS). Patients with episodic CH had a significantly higher 15-minute response rate with nVNS than those with chronic CH, at 34.2% and 13.6%, respectively (Figure).
Figure. Response rate (ITT population).
CH, cluster headache; ITT, intent-to-treat; nVNS, noninvasive vagus nerve stimulation
The rate of sustained-treatment response was significantly higher with nVNS compared with sham treatment. The bulk of the positive sustained response to nVNS was in the episodic cohort, which had a 34.2% sustained-treatment response rate versus 10.6% for sham treatment, whereas the respective rates for the chronic-CH cohort were 13.6% and 15.4%.
Patients with episodic CH responded in larger numbers than those with chronic CH both initially and over the first hour. Furthermore, there were no serious device-related adverse events. However, calculations of the rate of blinding at the end of the randomized phase “indicated that true blinding had not been achieved,” the authors noted in the accompanying poster.
—Rosemary Frei, MSc